|
8/3/2023 0 Comments Abbott istat fda approvalThe cartridge is then inserted into the handheld instrument.Įvaluating brain injuries is complex – and research shows that we only catch about half of those who show up to the hospital with a suspected TBI Geoffrey ManleyĪbbott is also working on a whole blood test, which would eliminate the need for separation of plasma and could be used at the patient's side in a healthcare setting. The test requires a small blood sample drawn from the arm, from which plasma is extracted with a centrifuge and applied to the test's cartridge. "You can't treat what you don't know and now physicians will be equipped with critical, objective information that will help them provide the best care possible, allowing patients to take steps to recover, prevent reinjury and get back to doing the things they care about most." "Healthcare providers have been waiting for a blood test for the brain and now we have one," said Beth McQuiston, M.D., medical director for Abbott's diagnostics business. For those who test positive, this test result complements CT scans to help clinicians evaluate whether someone has a TBI. A negative result on this test can be used to rule out the need for a head CT scan, a common tool used to diagnose concussion. This test measures specific proteins present in the blood after a TBI. TBIs, including concussions, are an alteration in brain function caused by an external force. Tests results are available within 15 minutes after plasma is placed in the test cartridge. For i-STAT test cartridge information and intended use, refer to individual product pages or the cartridge information (CTI/IFU) in the i-STAT Support area.Ībbott - A Leader in Rapid Point-of-Care Diagnostics.The test will run on Abbott's handheld i-STAT Alinity platform. 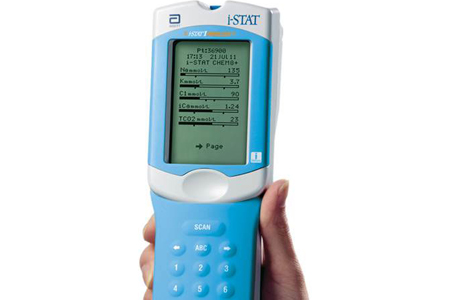
Check with your local representative for availability in specific markets. Not all products are available in all regions. GDPR Statement | Declaration for California Compliance Law. Any person depicted in such photographs is a model. Photos displayed are for illustrative purposes only. 
Your use of this website and the information contained herein is subject to our Website Terms and Conditions and Privacy Policy. 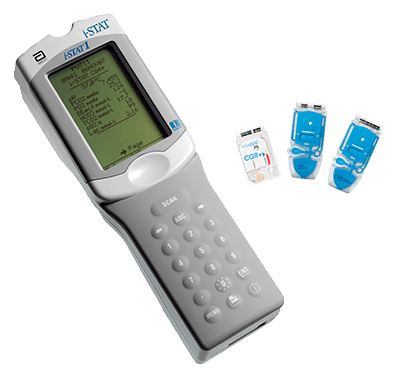
The products and information contained herewith may not be accessible in all countries, and Abbott takes no responsibility for such information which may not comply with local country legal process, regulation, registration and usage. This website is governed by applicable U.S. No use of any Abbott trademark, trade name, or trade dress in this site may be made without the prior written authorization of Abbott, except to identify the product or services of the company. Unless otherwise specified, all product and service names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates. This page is only accessible to registered i-STAT customers. 
Reference ranges (sometimes referred to as normal ranges) in the default Customization profile can be found behind login on the i-STAT Cartridge and Test Information (CTI) Sheets/Instructions for Use (IFU) page.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
